NCERT Solutions for Class 11th BIOLOGY
Chapter 11 Respiration in Plants
1. Give the schematic representation of an overall view of Krebs’ cycle.
Solution:

Solution:

2. Differentiate between
(a) Respiration and Combustion
(b) Glycolysis and Krebs’cycle
(c) Aerobic respiration and Fermentation
Solution: (a) Differences between respiration and combustion are as follows :

(b) Differences between glycolysis and Krebs’ cycle are as follows:


(C)Differences between aerobic respiration and fermentation are as follows:

(a) Respiration and Combustion
(b) Glycolysis and Krebs’cycle
(c) Aerobic respiration and Fermentation
Solution: (a) Differences between respiration and combustion are as follows :

(b) Differences between glycolysis and Krebs’ cycle are as follows:


(C)Differences between aerobic respiration and fermentation are as follows:

3. What are respiratory substrates? Name the most common respiratory substrate.
Solution: Respiratory substrates are those organic substances which are oxidised during respiration to liberate energy inside the living cells. The common respiratory substrates are carbohydrates, proteins, fats and organic acids. The most common respiratory substrate is glucose. It is a hexose monosaccharide.
Solution: Respiratory substrates are those organic substances which are oxidised during respiration to liberate energy inside the living cells. The common respiratory substrates are carbohydrates, proteins, fats and organic acids. The most common respiratory substrate is glucose. It is a hexose monosaccharide.
4. Give the schematic representation of glycolysis.
Solution:


5. Explain ETS.
Solution: An electron transport chain or system (ETS) is a series of coenzymes and cytochromes that take part in the passage of electrons from
a chemical to its ultimate acceptor. Reduced coenzymes participate in electron transport chain. Electron transport takes place on cristae of mitochondria [oxysomes ( F0 -F1 , particles) found on the inner surface of the membrane of mitochondria]. NADH formed in glycolysis and citric acid cycle are oxidised by NADH dehydrogenase (complex I) and the electrons are transferred to ubiquinone. Ubiquinone also receives reducing equivalents via FADH2 through the activity of succinate dehydrogenase (complex II). The reduced ubiquinone is then oxidised by transfer of electrons of cytochrome c via cytochrome Fc, complex (complex III). Cytochrome c acts as a mobile carrier between complex III and complex IV. Complex IV refers to cytochrome c oxidase complex containing cytochromes a and a3and two copper centres. When the electrons are shunted over the carriers via complex I to IV in the electron transport chain, they are coupled to ATP synthetase (complex V) for the formation of ATP from ADP and Pi. Oxygen functions as the terminal acceptor of electrons and is reduced to water along with the hydrogen atoms. Reduced coenzymes (coenzyme I, II and FAD) do not combine directly with the molecular O2. Only their hydrogen or electrons are transferred through various substances and finally reach O2. The substances useful for the transfer of electron are called electron carriers. Only electrons are transferred through cytochromes (Cyt F1 Cyt c,,C2, a, a3) and finally reach molecular O2. Both cytochrome a and a3 form a system called cytochrome oxidase. Copper is also present in Cyt a3 in addition to iron. The molecular oxygen that has accepted electrons now receives the protons that were liberated into the surrounding medium to give rise to a molecule of water. The liberated energy is utilised for the synthesis of ATP from ADP and Pi.
Solution:


5. Explain ETS.
Solution: An electron transport chain or system (ETS) is a series of coenzymes and cytochromes that take part in the passage of electrons from
a chemical to its ultimate acceptor. Reduced coenzymes participate in electron transport chain. Electron transport takes place on cristae of mitochondria [oxysomes ( F0 -F1 , particles) found on the inner surface of the membrane of mitochondria]. NADH formed in glycolysis and citric acid cycle are oxidised by NADH dehydrogenase (complex I) and the electrons are transferred to ubiquinone. Ubiquinone also receives reducing equivalents via FADH2 through the activity of succinate dehydrogenase (complex II). The reduced ubiquinone is then oxidised by transfer of electrons of cytochrome c via cytochrome Fc, complex (complex III). Cytochrome c acts as a mobile carrier between complex III and complex IV. Complex IV refers to cytochrome c oxidase complex containing cytochromes a and a3and two copper centres. When the electrons are shunted over the carriers via complex I to IV in the electron transport chain, they are coupled to ATP synthetase (complex V) for the formation of ATP from ADP and Pi. Oxygen functions as the terminal acceptor of electrons and is reduced to water along with the hydrogen atoms. Reduced coenzymes (coenzyme I, II and FAD) do not combine directly with the molecular O2. Only their hydrogen or electrons are transferred through various substances and finally reach O2. The substances useful for the transfer of electron are called electron carriers. Only electrons are transferred through cytochromes (Cyt F1 Cyt c,,C2, a, a3) and finally reach molecular O2. Both cytochrome a and a3 form a system called cytochrome oxidase. Copper is also present in Cyt a3 in addition to iron. The molecular oxygen that has accepted electrons now receives the protons that were liberated into the surrounding medium to give rise to a molecule of water. The liberated energy is utilised for the synthesis of ATP from ADP and Pi.
7: Distinguish between the following:
(a) Aerobic respiration and Anaerobic respiration
(b) Glycolysis and Fermentation
(c) Glycolysis and Citric acid Cycle
Answer
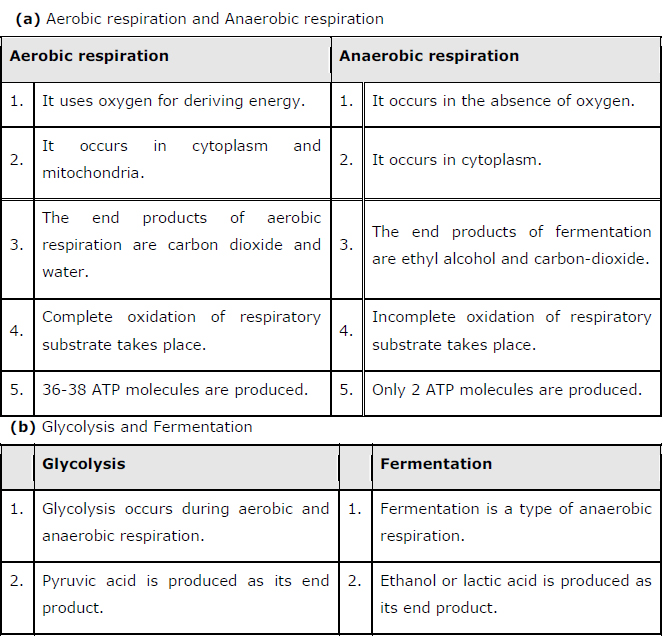

(a) Aerobic respiration and Anaerobic respiration
(b) Glycolysis and Fermentation
(c) Glycolysis and Citric acid Cycle
Answer


Question 8: What are the assumptions made during the calculation of net gain of ATP?
Answer For theoretical calculation of ATP molecules, various assumptions are made, which are as follows.
(a) It is assumed that various parts of aerobic respiration such as glycolysis, TCA cycle, and ETS occur in a sequential and orderly pathway.
(b) NADH produced during the process of glycolysis enters into mitochondria to undergo oxidative phosphorylation.
(c) Glucose molecule is assumed to be the only substrate while it is assumed that no other molecule enters the pathway at intermediate stages.
(d) The intermediates produced during respiration are not utilized in any other process.
Answer For theoretical calculation of ATP molecules, various assumptions are made, which are as follows.
(a) It is assumed that various parts of aerobic respiration such as glycolysis, TCA cycle, and ETS occur in a sequential and orderly pathway.
(b) NADH produced during the process of glycolysis enters into mitochondria to undergo oxidative phosphorylation.
(c) Glucose molecule is assumed to be the only substrate while it is assumed that no other molecule enters the pathway at intermediate stages.
(d) The intermediates produced during respiration are not utilized in any other process.
Question 9: Discuss “The respiratory pathway is an amphibolic pathway.”
Answer Respiration is generally assumed to be a catabolic process because during respiration, various substrates are broken down for deriving energy. Carbohydrates are broken down to glucose before entering respiratory pathways. Fats get converted into fatty acids and glycerol whereas fatty acids get converted into acetyl CoA before
entering the respiration. In a similar manner, proteins are converted into amino acids, which enter respiration after deamination. During synthesis of fatty acids, acetyl CoA is withdrawn from respiratory pathway. Also, in the synthesis of proteins, respiratory substrates get withdrawn. Thus, respiration is also involved in anabolism. Therefore, respiration can be termed as amphibolic pathway as it involves both anabolism and catabolism.
Answer Respiration is generally assumed to be a catabolic process because during respiration, various substrates are broken down for deriving energy. Carbohydrates are broken down to glucose before entering respiratory pathways. Fats get converted into fatty acids and glycerol whereas fatty acids get converted into acetyl CoA before
entering the respiration. In a similar manner, proteins are converted into amino acids, which enter respiration after deamination. During synthesis of fatty acids, acetyl CoA is withdrawn from respiratory pathway. Also, in the synthesis of proteins, respiratory substrates get withdrawn. Thus, respiration is also involved in anabolism. Therefore, respiration can be termed as amphibolic pathway as it involves both anabolism and catabolism.
Question 10: Define RQ. What is its value for fats?
Answer Respiratory quotient (RQ) or respiratory ratio can be defined as the ratio of the volume of CO2 evolved to the volume of O2 consumed during respiration. The value of respiratory quotient depends on the type of respiratory substrate. Its value is one for carbohydrates. However, it is always less than one for fats as fats consume more oxygen for respiration than carbohydrates. It can be illustrated through the example of tripalmitin fatty acid, which consumes 145 molecules of O2 for respiration while 102 molecules of CO2 are evolved. The RQ value for tripalmitin is 0.7.
Answer Respiratory quotient (RQ) or respiratory ratio can be defined as the ratio of the volume of CO2 evolved to the volume of O2 consumed during respiration. The value of respiratory quotient depends on the type of respiratory substrate. Its value is one for carbohydrates. However, it is always less than one for fats as fats consume more oxygen for respiration than carbohydrates. It can be illustrated through the example of tripalmitin fatty acid, which consumes 145 molecules of O2 for respiration while 102 molecules of CO2 are evolved. The RQ value for tripalmitin is 0.7.
11. What is oxidative phosphorylation?
Solution: Oxidative phosphorylation is the synthesis of energy rich ATP molecules with the help of energy liberated during oxidation of reduced co-enzymes (NADH, FADH2) produced in respiration. The enzyme required for this synthesis is called ATP synthase. It is considered to be the fifth complex of electron transport chain. ATP synthase is located in FT or head piece of F0 -F1 or elementary particles. The particles are present in the inner mitochondrial membrane. ATP synthase becomes active in ATP formation only where there is a proton gradient having higher concentration of H+ or protons on the F0 side as compared to F x side (chemiosmotic hypothesis of Peter Mitchell).
Increased proton concentration is produced in the outer chamber or outer surface of inner mitochondrial membrane by the pushing of proton with the help of energy liberated by passage of electrons from one carrier to another. Transport of the electrons from NADH over ETC helps in pushing three pairs of protons to the outer chamber while two pairs of protons are sent outwardly during electron flow from FADH2. The flow of protons through the F0 channel induces F1 particle to function as ATP-synthase. The energy of the proton gradient is used in attaching a phosphate radical to ADP by high energy bond. This produces ATP. Oxidation of one molecule of NADH2 produces 3 ATP molecules while a similar oxidation of FADH2 forms 2 ATP molecules.
Solution: Oxidative phosphorylation is the synthesis of energy rich ATP molecules with the help of energy liberated during oxidation of reduced co-enzymes (NADH, FADH2) produced in respiration. The enzyme required for this synthesis is called ATP synthase. It is considered to be the fifth complex of electron transport chain. ATP synthase is located in FT or head piece of F0 -F1 or elementary particles. The particles are present in the inner mitochondrial membrane. ATP synthase becomes active in ATP formation only where there is a proton gradient having higher concentration of H+ or protons on the F0 side as compared to F x side (chemiosmotic hypothesis of Peter Mitchell).
Increased proton concentration is produced in the outer chamber or outer surface of inner mitochondrial membrane by the pushing of proton with the help of energy liberated by passage of electrons from one carrier to another. Transport of the electrons from NADH over ETC helps in pushing three pairs of protons to the outer chamber while two pairs of protons are sent outwardly during electron flow from FADH2. The flow of protons through the F0 channel induces F1 particle to function as ATP-synthase. The energy of the proton gradient is used in attaching a phosphate radical to ADP by high energy bond. This produces ATP. Oxidation of one molecule of NADH2 produces 3 ATP molecules while a similar oxidation of FADH2 forms 2 ATP molecules.
12. What is the significance of step-wise release of energy in respiration?
Solution: The utility of step-wise release of energy in respiration are given as follows :
(i) There is a step-wise release of chemical bond energy which is very easily trapped in forming ATP molecules.
(ii) Cellular temperature is not allowed to rise.
(iii) Wastage of energy is reduced.
(iv) There are several intermediates which can be used in production of a number of biochemicals.
(v) Through their metabolic intermediates different substances can undergo respiratory catabolism.
(vi) Each step of respiration is controlled by its own enzyme. The activity of different enzymes can be enhanced or inhibited by specific compounds.
This helps in controlling the rate of respiration and the amount of energy liberated by it.
Solution: The utility of step-wise release of energy in respiration are given as follows :
(i) There is a step-wise release of chemical bond energy which is very easily trapped in forming ATP molecules.
(ii) Cellular temperature is not allowed to rise.
(iii) Wastage of energy is reduced.
(iv) There are several intermediates which can be used in production of a number of biochemicals.
(v) Through their metabolic intermediates different substances can undergo respiratory catabolism.
(vi) Each step of respiration is controlled by its own enzyme. The activity of different enzymes can be enhanced or inhibited by specific compounds.
This helps in controlling the rate of respiration and the amount of energy liberated by it.

Comments
Post a Comment